Medical Information Database Research and Investigation
About Us
A specialized team with the necessary skills for database research and surveys will provide full support from planning to reporting.
Surveys and research using databases require the review of various experts. We have established an operational system with a department specialized in database surveys and research and provide a series of support services.
EPS Features
Expertise
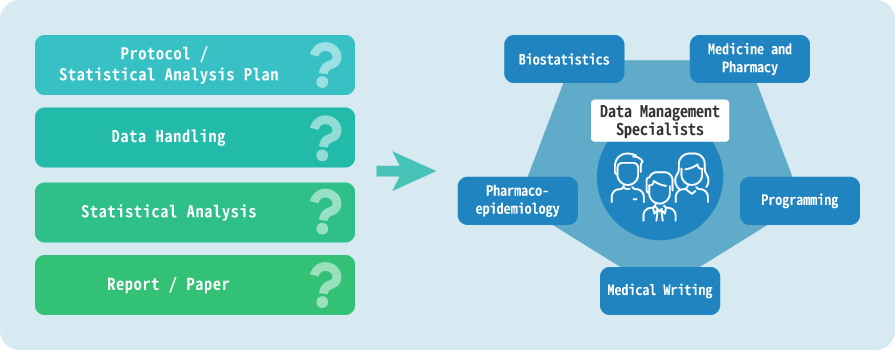
With staff experienced in the medical field, we establish an operational structure in a department specialized in database survey and research.
Comprehensive Support
Our experts provide comprehensive support for planning, protocol creation, statistical analysis planning and implementation, and report creation.
Extensive Achievements
- Proven track record with a variety of databases.
- Extensive experience in the whole process, from planning to reporting.
Services
Research Questions that can be reviewed in the database
- Estimate the magnitude of the difference between the control drug and the drug for a specific safety risk.
- Impact of covariates on specific safety risks.
(*Risk analysis in particular groups such as infants, elders, decreased liver, and kidney function.) - Evaluation of changes in the frequency of occurrence of specific adverse events before and after the implementation of safety measures, etc.
Main Services
- Database survey and protocol creation support
- Database survey and study implementation deliberation
- Database selection
- Study design selection
- Validation studies planning and implementation
- Statistical analysis planning and implementation
- Extraction of target diseases conditions
- Examination of conditions for deriving exposure variables, covariate variables, and outcome variables (including preparation of code lists for extraction)
- Report creation support
- Theory copywrites submission support